|
2 The 4 Technology Solutions

NATURAL ATTENUATION PART V:
METHANOGENIC SYSTEMS
An On-Line Version of a Column First Published in:
Environmental Technology March/April 19967 Vol. 7 No. 2
by: David B. Vance dbv7@mindspring.com
Previous Groundwater Columns have covered various aspects of "Natural Attenuation". Basic principles and processes, the effect natural electron acceptors in pump and treat systems, and the dynamics of natural groundwater flow systems incorporating the effects of dispersion. This column will discuss the ultimate of no action alternatives; the use of methanogenic microbial systems (where the electron acceptor is the contaminating hydrocarbon) to degrade petroleum and xenobiotic hydrocarbons.
First, it is important to understand the difference between Methanotrophic bacteria and methanogenic bacteria. Methanotrophs use oxygen to oxidize methane into carbon dioxide (CO2). Methanotrophic bacterial systems have received a great deal of attention over the last ten years since it has been found that methane mono oxygenase (the enzyme generated by Methanotrophs to react with methane) can degrade a wide variety of chlorinated hydrocarbons. The process is known as co-metabolism and is definitely an aerobic process.
Methanogenesis is the process of degrading hydrocarbons with the end product being methane (CH4) gas and carbon dioxide.
The general reaction is as follows:
- 2Corganic + 2H2O = CO2 + CH4
This is a strictly anaerobic process, methanogenic bacteria are poisoned by the presence of oxygen at levels as low as 0.18 mg/L of soluble oxygen (as O2).
The redox conditions under which these two different microbial systems operate are literally at opposite ends of the spectrum:
- Methanotrophic reactions occur at the Eh range of + 250 mV
- Methanogenic reactions occur at the Eh range of - 200 mV.
As an aside, methanogenic bacteria are one of the three classes of bacteria termed Archaebacteria, which are representative of organisms that first appeared on Earth some 3.5 billion years ago.
While their activity is inhibited by oxygen, these bacteria are robust enough to appear in a wide variety of natural locations such as: the intestinal tracts of ruminant mammals (cows etc.), sewage digesters, groundwater and soil.
The precise mechanisms of hydrocarbon degradation under methanogenic conditions is not entirely understood by current researchers. Theoretically, methanogenic reactions only involve chemicals containing one or two carbons. More complex hydrocarbons are degraded through the synergistic activity of other bacteria that may utilize by products (such as hydrogen) of the methanogenic process. Definitive illumination of these processes has not occurred in the laboratory, as it takes rigorous physical controls as well as a great deal of skill to culture these bacteria. To date no one has been able to grow (in the lab) a complete consortia of degrading anaerobes as conjectured above.
With regards to soils and groundwater, Figure 1 serves to illustrate that methanogenesis does occur. The data presented in the figure was collected using soil gas above contaminated groundwater systems from diverse geographic locations (Chicago, Indiana, Tennessee, and Oklahoma).
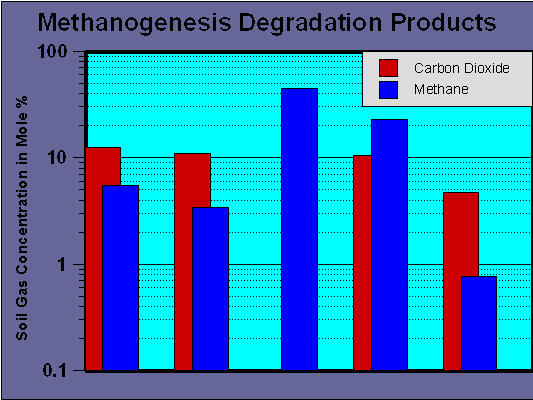
Figure 1 - Soil Gas Methane and Carbon Dioxide From Methanogenic Hydrocarbon Degradation
The concentrations of methane and carbon dioxide are expressed as mole percent. This data is somewhat remarkable since this should be a difficult reaction to initiate. Oxygen, nitrate, and sulfate are all toxic or inhibitory to methanogenic activity. That means that at any site at which evidence of methanogenesis is present (methane gas) there has been a series of biodegradation reactions that have consumed the alternative electron acceptors. Migrating from the exterior to the interior of a plume, the type of redox conditions (Eh) will change from + 250 Mv to - 200 Mv, with oxygen, nitrate, and sulfate progressively being consumed.
The degradation of chlorinated xenobiotic compounds under methanogenic conditions is particularly enhanced. Not from the direct action of the bacteria, but from the presence of molecular hydrogen that is produced from water during the methanogenic process. A previous column reviewed the mechanisms that have been discovered over the last few years for the dehalogenation of chlorinated hydrocarbons, one of which includes the incorporation of hydrogen under methanogenic conditions.
Now back to the statement in the introduction of this column, of methanogenesis being "the ultimate of no action alternatives". The physical/chemical requirement for the removal of all other potential electron acceptors infers that the hydrodynamics of such a system are relatively quiescent. The transport of alternate electron acceptors into the core of the contamination plume must be at a rate slow enough to allow for the consumption of all alternate electron acceptors before the methanogenic core zone is reached.
This would be a positive argument for absolutely no action involving pump and treat systems. That would increase groundwater velocities, introducing inhibitory electron acceptors into the active methanogenic zone. Groundwater systems in fine grained soils, where transport properties are poor, would be ideal for the exploitation of methanogenic degradation.
Of course, one problem with methanogenic degradation is the kinetics of the process. Methanogenic degradation occurs at rates that are orders of magnitude slower than the rates seen with other electron acceptors. However, at some sites with poor transport conditions or inaccessibility (i.e. a deep groundwater table) this would still be a reasonable alternative.
Unfortunately there are other problems that are far more significant. The number and types of hydrocarbons degraded under methanogenic conditions is very limited. Some laboratory studies have demonstrated the methanogenic degradation of toluene and o-xylene with no degradation of m-xylene, p-xylene, ethylbenzene, or benzene. Others have found evidence of degradation of benzene with recalcitrance towards other compounds.
The data from Figure 1 and other sources indicates that methanogenesis is readily activated through natural processes in the core of a contaminant plumes. Unfortunately, the process appears to be extremely selective and not capable of the complete degradation of all contaminants present in a typical hydrocarbon plume. Under instances where a specific compound has been released (such as toluene used as a solvent) methanogenesis may be a viable natural attenuation process. It also has value in the natural dehalogenation of chlorinated compounds.
In instances where an aquifer has good transport qualities and a supply of natural electron acceptors, natural attenuation alone will be viable process and acceptable to the regulatory community. In other cases supplementation of electron acceptors or improvement of groundwater dynamics through pump and treat may provide an adequate minimal approach. But, in most instances methanogenesis as the "ultimate of no action alternatives" is not going to be applicable.
Return to the Environmental Technology Index Page
Copyright 2008 David B. Vance
All Rights Reserved

If you have comments or suggestions, e-mail me at dbv7@mindspring.com
|